Kampala, Uganda | THE INDEPENDENT | As a result of the changing findings into treatment of the Coronavirus (COVID-19), the Ministry of Health is set to change their treatment protocols to match emerging evidence.
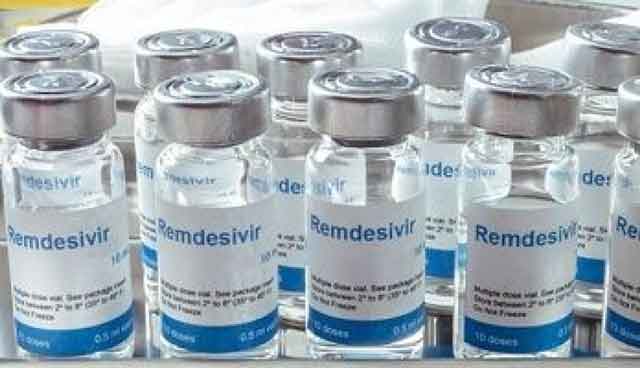
Dr. William Worodria who heads the National COVID-19 case management team told Uganda Radio Network – URN that they have changed their guidelines twice before and they quickly get obsolete with new research. He says they are currently compiling the list of new therapies but they will not include popular antiviral drug remdesivir which was approved for treatment by the US Food and Drug Administration last week.
The Ebola drug whose use among COVID-19 patients has been controversial globally in recent weeks with the World Health Organization declaring it has little benefit in treatment had been planned for roll out in Uganda in September following and two Indian pharmaceutical companies including CIPLA that has a subsidiary in the country and Heteropharma were negotiating for supply deals.
Dr. Bruce Kirenga, another member of the case management team had earlier told URN that one of the companies had ferried some doses of the drug into the country awaiting health workers to complete training on how to deliver the intravenously given drug before roll out could happen.
But, now, Dr. Worodria says it would be very expensive for Uganda to roll the drug out.
For him, if they were to roll it out here, it would be accessed by people who are ready to pay for it since a dose costs about $150 (UGX553,000) and yet a person needs to be on medication for five to ten days.
Dr. Worodria further explains that the drug can have an effect once given early into infection for instance in a week since it works by blocking the ability of the virus to reproduce and weaken the body.
However, in many cases in Uganda in the first week of infection, patients are not yet identified as the majority get tested when they develop symptoms of the disease.
For him, for this drug to be adopted in their treatment guidelines considering the cost – benefit implications associated they need more solid evidence attributed to it.
The drug originally manufactured by global pharmaceutical Gilead Sciences is given intravenously and has been rolled out for treatment of COVID-19 in many countries including neighboring Kenya. The other countries using the drug include; USA, United Kingdom, Japan, South Korea, Taiwan, Singapore, Israel, Australia and India.
Initial research showed that the drug can cut hospital stay for admitted patients by four to five days, a reason as to why many countries chose to use it as an emergency drug.
********
URN
 The Independent Uganda: You get the Truth we Pay the Price
The Independent Uganda: You get the Truth we Pay the Price